RESEARCH
From natural insulins to designed analogs
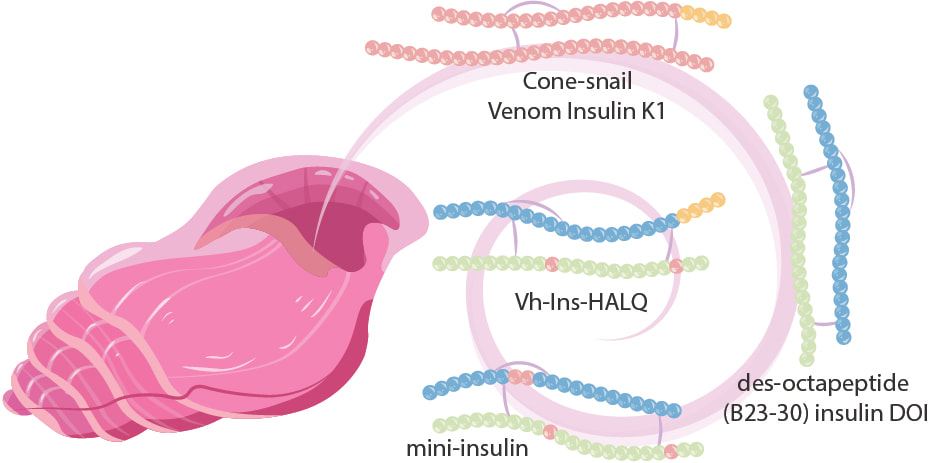
Although currently available insulin analogues reduce blood glucose levels, this blood glucose lowering action is not regulated in a glucose dependent fashion. The major consequence of excess insulin administration is hypoglycemia, which can lead to acute complications such as loss of consciousness, coma, and even death. On the other hand, insufficient insulin administration leads to chronic hyperglycemia and complications such as blindness, kidney failure, and heart disease. There remains a need to modulate the kinetics of injected insulin therapy to more closely match the dynamics of fluctuating blood glucose levels. We are interested in developing glucose-responsive or “smart” insulin with its biological activity regulated by circulating glucose levels. Ideally, smart insulin would be administered once daily and, by responding to the circulating local glucose levels, would maintain normal glucose levels throughout the day. Furthermore, we are developing ultrafast-acting insulin with fast-on, fast-off properties for uses in artificial pancreas. Recently, we developed antagonistic insulin derivatives, which block insulin signaling with high potencies. We are currently investigating their roles in treating hyperinsulinism.
Although currently available insulin analogues reduce blood glucose levels, this blood glucose lowering action is not regulated in a glucose dependent fashion. The major consequence of excess insulin administration is hypoglycemia, which can lead to acute complications such as loss of consciousness, coma, and even death. On the other hand, insufficient insulin administration leads to chronic hyperglycemia and complications such as blindness, kidney failure, and heart disease. There remains a need to modulate the kinetics of injected insulin therapy to more closely match the dynamics of fluctuating blood glucose levels. We are interested in developing glucose-responsive or “smart” insulin with its biological activity regulated by circulating glucose levels. Ideally, smart insulin would be administered once daily and, by responding to the circulating local glucose levels, would maintain normal glucose levels throughout the day. Furthermore, we are developing ultrafast-acting insulin with fast-on, fast-off properties for uses in artificial pancreas. Recently, we developed antagonistic insulin derivatives, which block insulin signaling with high potencies. We are currently investigating their roles in treating hyperinsulinism.
Modulating interleukins activities in vivo
Interleukins are cytokines that can either stimulate or inhibit T cell responses. Interleukin-2 (IL-2), for instance, is a multifunctional cytokine that can bolster or suppress immune reactions depending on the type of target cell involved. In recent years, protein engineering of IL-2 has garnered significant interest with the goal of creating improved IL-2 therapeutics boasting superior safety profiles. Using protein chemistry approaches, we design protein-based therapeutics furnished with switches to control bioactivity. We project that these switch-regulated IL-2 agents will have considerable advantages over existing candidates, which primarily aim to bind to dimeric IL-2Rs rather than the trimeric receptors. Our goal is to create a platform wherein distinct switches can be incorporated into IL-2 to yield specific cell-targeting properties. The IL-2 therapeutics will possess the benefits of both antibody-drug conjugates and pro-drugs, optimizing their therapeutic efficacy while curtailing off-target effects.
Interleukins are cytokines that can either stimulate or inhibit T cell responses. Interleukin-2 (IL-2), for instance, is a multifunctional cytokine that can bolster or suppress immune reactions depending on the type of target cell involved. In recent years, protein engineering of IL-2 has garnered significant interest with the goal of creating improved IL-2 therapeutics boasting superior safety profiles. Using protein chemistry approaches, we design protein-based therapeutics furnished with switches to control bioactivity. We project that these switch-regulated IL-2 agents will have considerable advantages over existing candidates, which primarily aim to bind to dimeric IL-2Rs rather than the trimeric receptors. Our goal is to create a platform wherein distinct switches can be incorporated into IL-2 to yield specific cell-targeting properties. The IL-2 therapeutics will possess the benefits of both antibody-drug conjugates and pro-drugs, optimizing their therapeutic efficacy while curtailing off-target effects.
Methodology Development for Creating New Peptides and Proteins
We are also developing new methods, both chemical and enzymatic, to generate peptides and proteins that are difficult to synthesize. First, we are involved in developing methods for peptide macrocyclizations, which are known to improve properties for peptide therapeutics. Furthermore, we are interested in methods to generate new insulin analogs and modified insulin derivatives. Last but not east, we are interested in using biopolymers to improve the formulation of peptides and proteins.
We are also developing new methods, both chemical and enzymatic, to generate peptides and proteins that are difficult to synthesize. First, we are involved in developing methods for peptide macrocyclizations, which are known to improve properties for peptide therapeutics. Furthermore, we are interested in methods to generate new insulin analogs and modified insulin derivatives. Last but not east, we are interested in using biopolymers to improve the formulation of peptides and proteins.